Tobacco artwork compliance,
made easy.
AI-assisted review against FDA CTP warnings, state packaging rules and in-house guides for faster, error-free label sign-off.


AI-assisted review against FDA CTP warnings, state packaging rules and in-house guides for faster, error-free label sign-off.


























Eliminate warning rotation errors, accelerate regulatory approvals.
AI-assisted checks against FDA CTP rules, TTB requirements, and brand guidelines to surface compliance issues early.
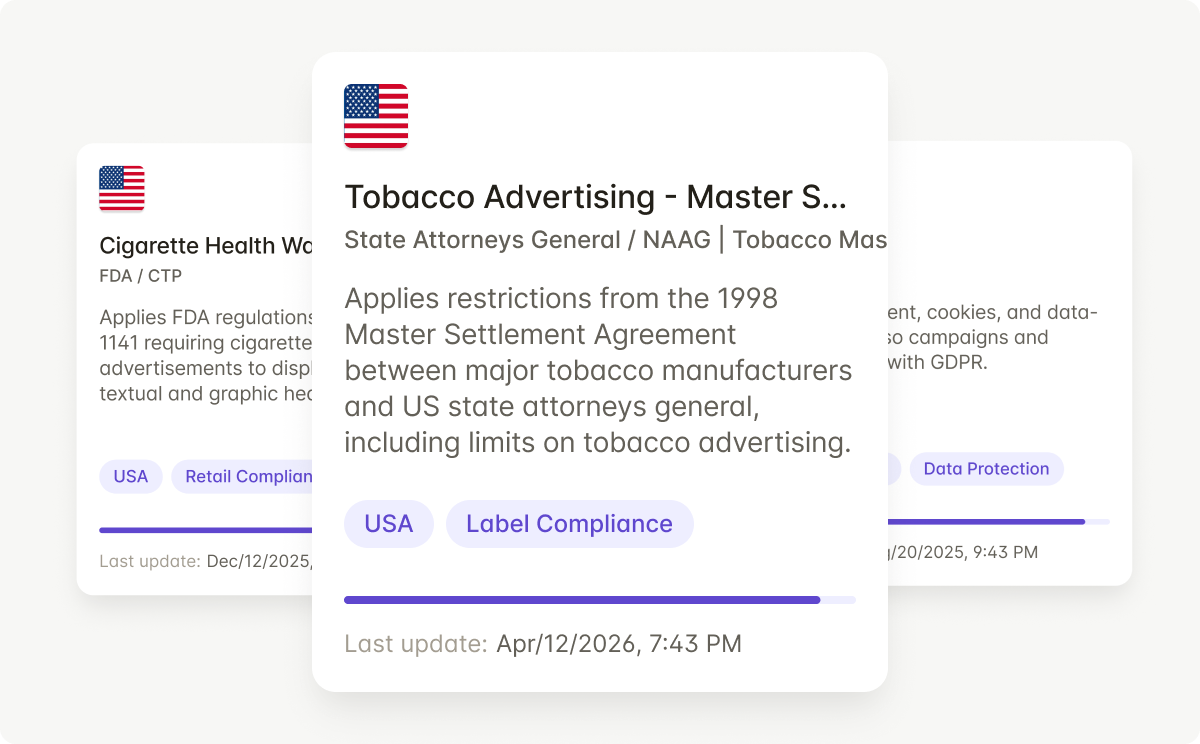
Rules automatically update when FDA CTP guidance changes or state packaging rules shift -- so every review reflects current regulations.
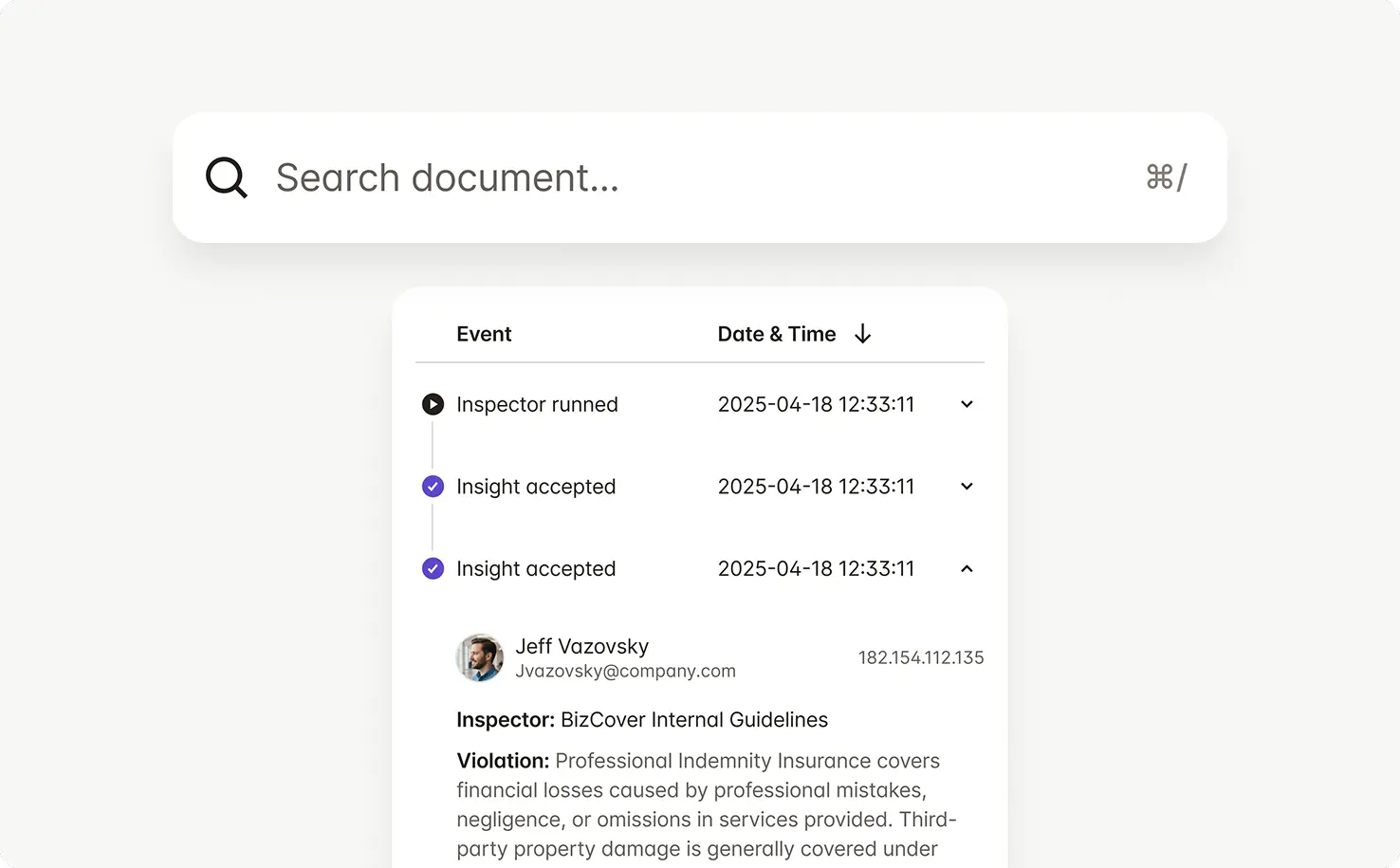
Every check, warning rotation, and approval is captured — creating a defensible record for FDA inspections, retailer audits, and internal review.

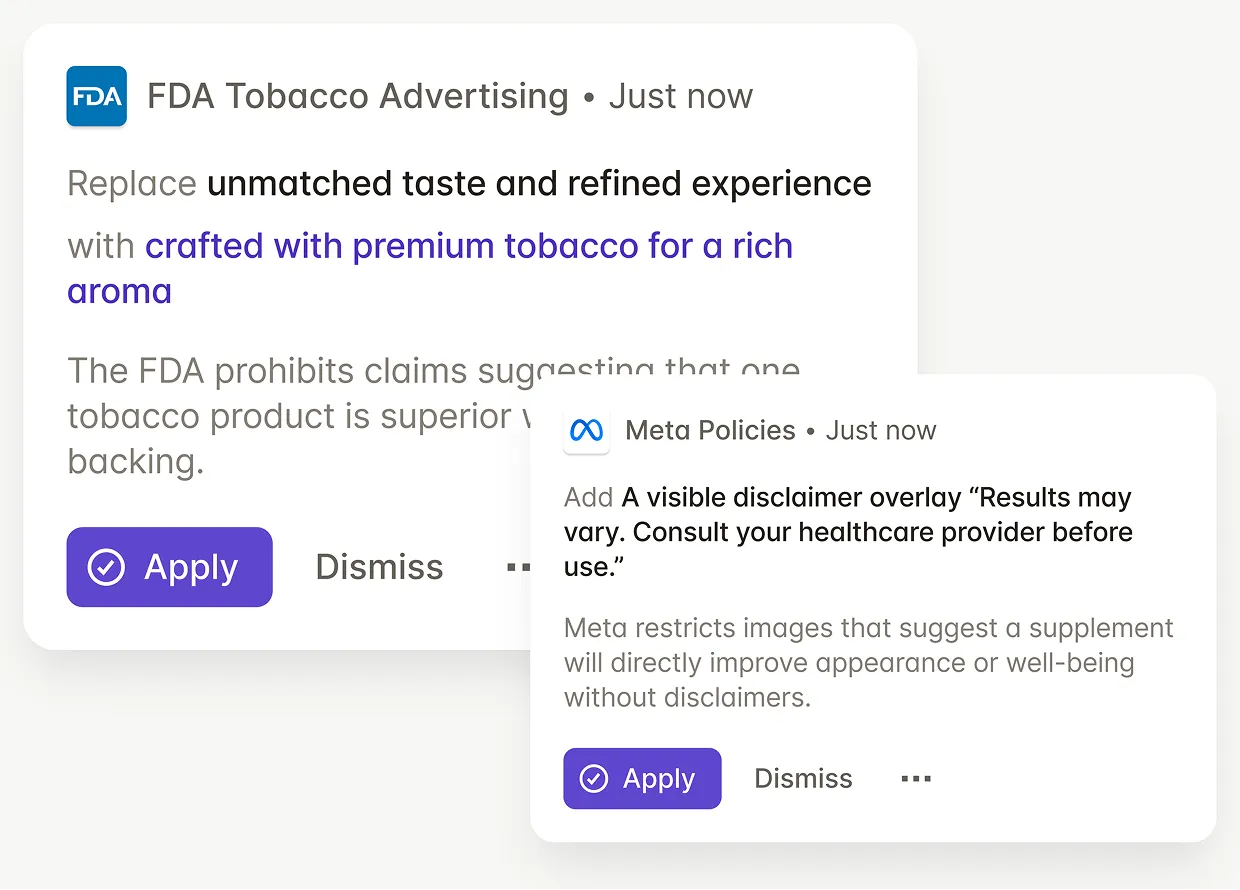
Check packaging artwork against FDA CTP requirements, state-level restrictions, and international market regulations -- one review, every market.
Start reviewing tobacco labels today. Free trial, no credit card required.
A shared review layer for tobacco packaging, used across regulatory, design, legal, and QA teams.
Design teams catch issues while working on artwork directly in design tools, reduce back-and-forth, and speed up alignment with marketing and operations.
These teams use GetGenAI to manage reviews across stages, handle multiple markets and SKUs, and keep label updates under control from brief to pre-print.

Legal and regulatory teams use GetGenAI to review packaging against up-to-date regulations and focus on complex cases instead of repetitive manual checks.

QA teams rely on structured review results and a full audit trail to confirm packaging is production-ready and reduce the risk of reprints or recalls.
Exploring AI, compliance, and tobacco packaging validation practices