Pharma artwork compliance, made easy.
AI-assisted checks across FDA 21 CFR Part 201, prescription drug labeling requirements, SPL standards, and in-house rules.
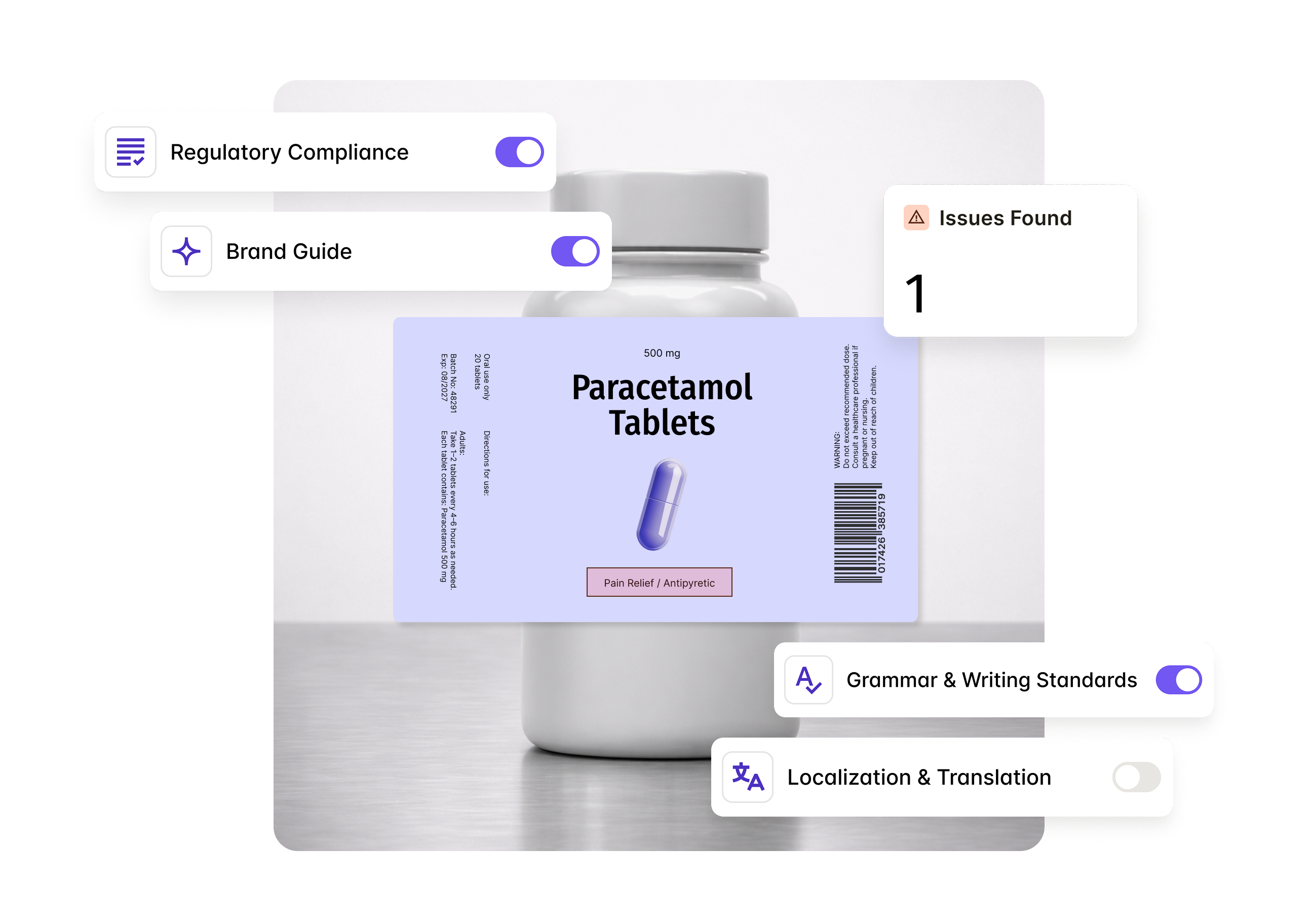

AI-assisted checks across FDA 21 CFR Part 201, prescription drug labeling requirements, SPL standards, and in-house rules.


























Eliminate human error, accelerate approvals.
AI-assisted checks across regulations and brand guidelines to surface issues early.
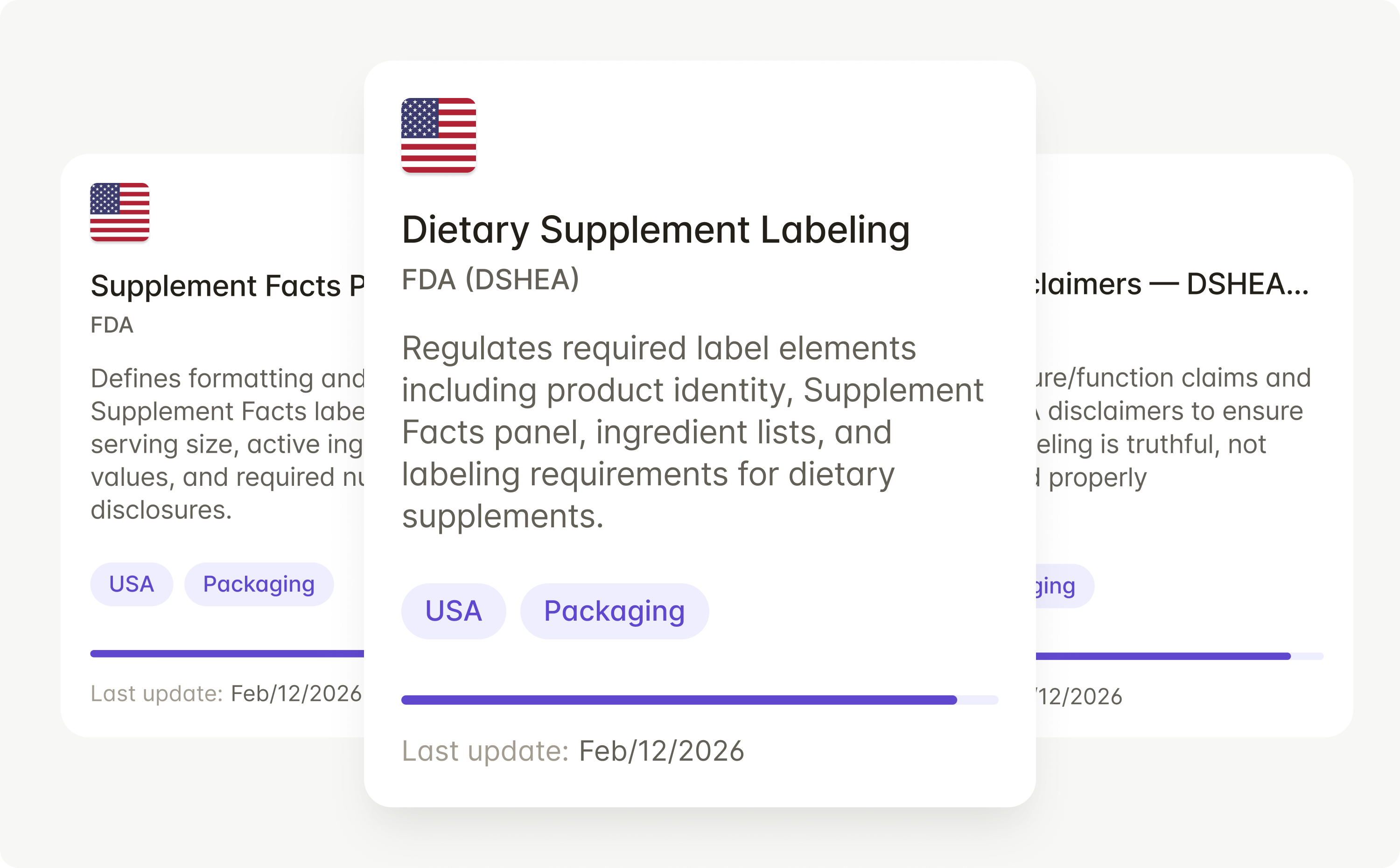
Rules automatically update across FDA 21 CFR Part 201, prescription and OTC drug labeling requirements, and SPL formatting standards.
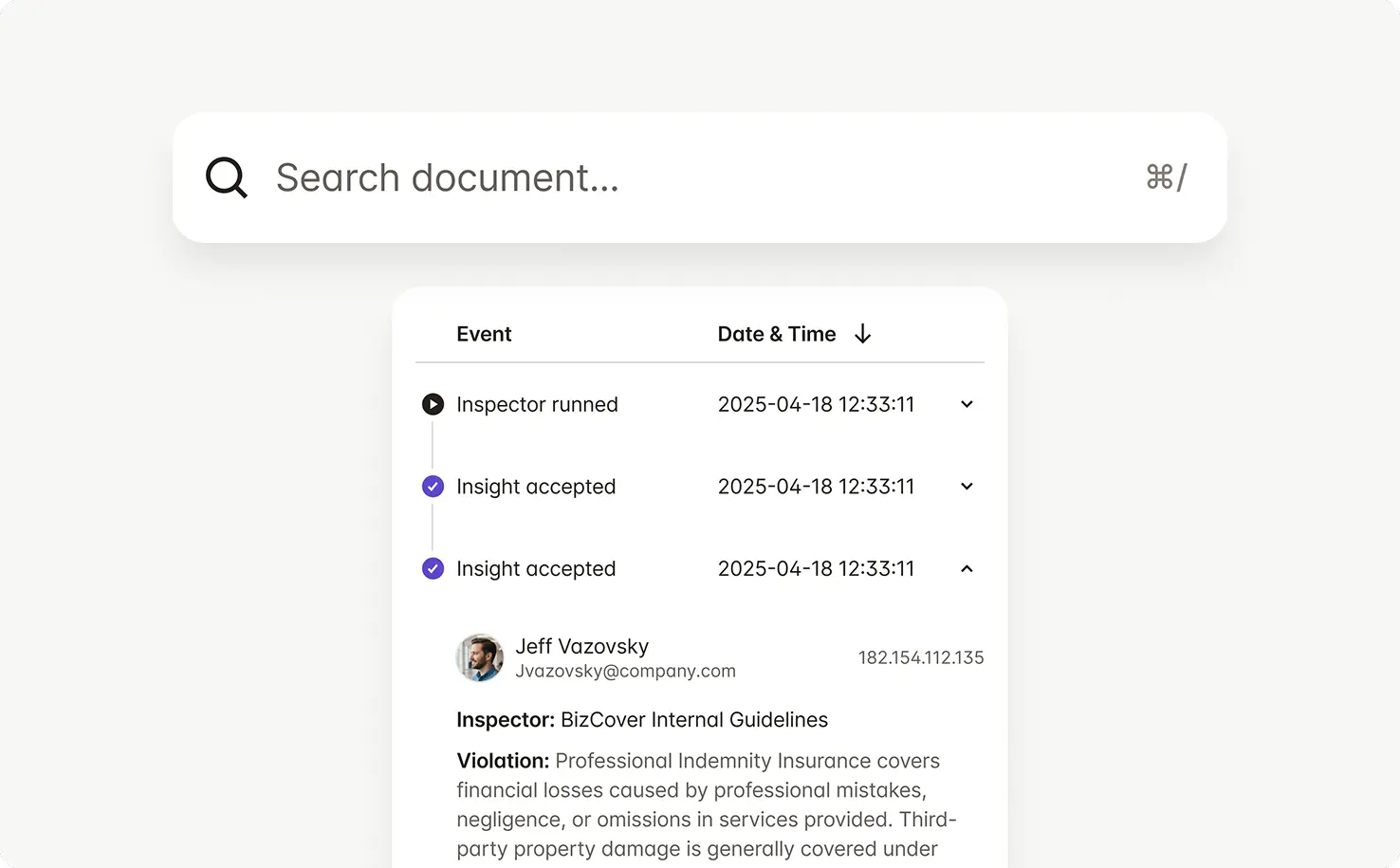
Every check, change, and approval is captured — showing what was reviewed, who approved it, and why.

Check packaging artwork against country-specific requirements for each market.
A shared review layer for packaging, used across roles, stages, and markets.
Founders review drug labels for claims, ingredient listings, and 21 CFR Part 201 requirements without relying fully on regulatory consultants.
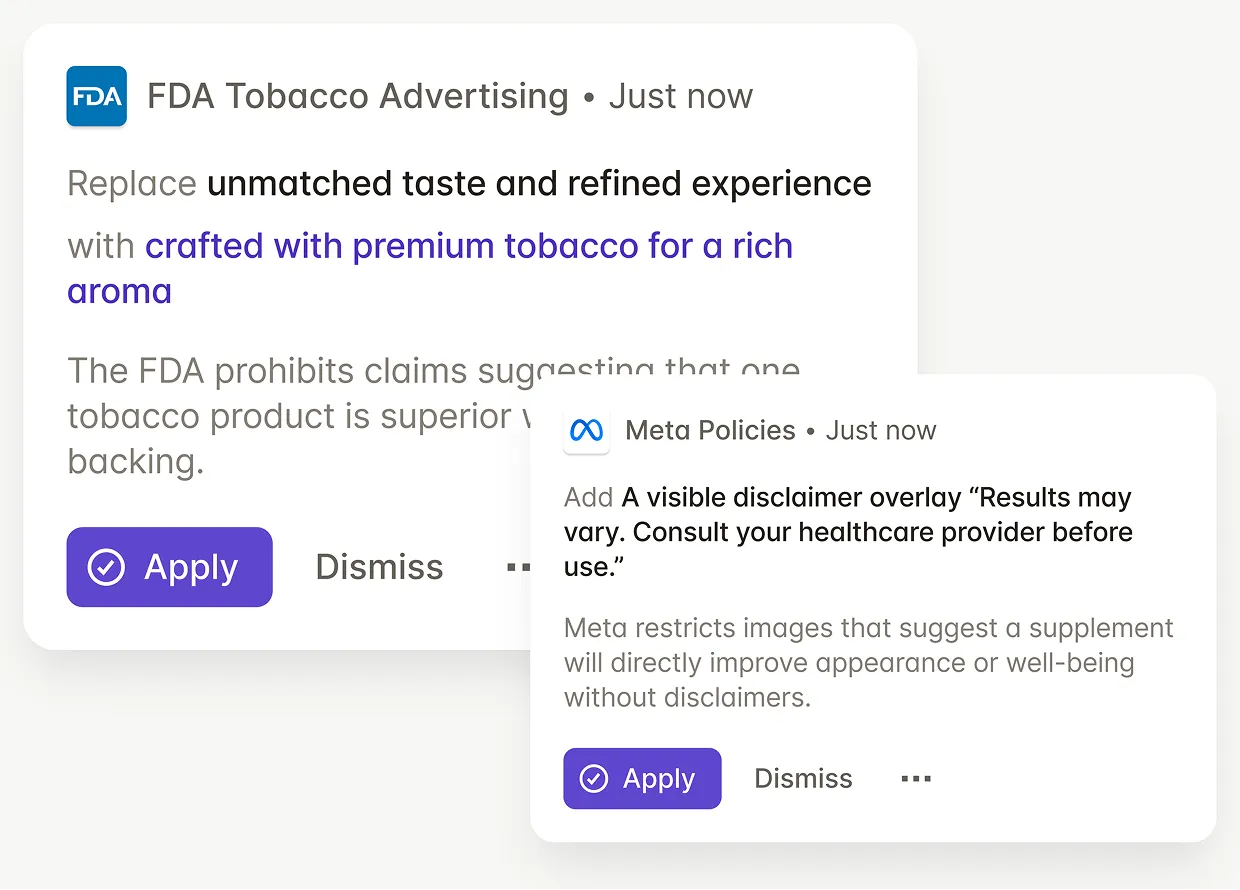
Regulatory teams check labels against auto-updating FDA prescription and OTC drug labeling requirements, reducing manual reviews and preventing non-compliant claims.

Operations teams manage label reviews across SKUs, keeping drug facts panels and ingredient listings aligned as formulations change.

Design teams catch Drug Facts formatting issues, warning statement placement errors, and layout inconsistencies before approval.
Start reviewing labels today. Free 7 day trial, no credit card required.
Exploring AI, compliance, and packaging validation practices